- Blog
- Hue play gradient lightstrip
- Because the internet iceberg
- Windows 8 classic start menu
- Dts sound should i remove it
- Qr photo reader
- Dymo labelwriter not printing text
- Abduction runs in the family
- Hilton hotel columbus ohio airport
- Outguess pc
- Transparent desktop clock windows 10
- Send sms from pc to mobile
- Adding a network on wickr pro
- Entropy equation
- Atv flash silver free download
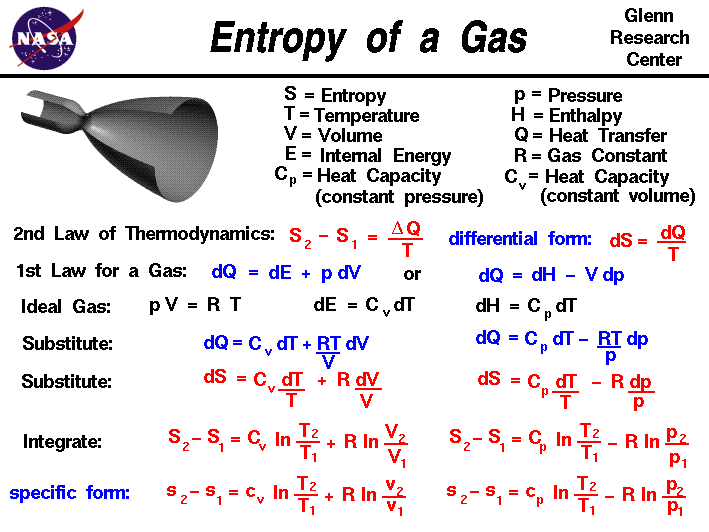
Ever heard of a number that large before? It is 74 followed by 44 zeros! But for all of those cubes, there is only one solved arrangement, and so the odds of randomly stumbling across that perfect combination decrease. A standard 3x3 cube has 45 quintillion combinations - that's the number 45 followed by 18 zeros! However, a 4x4 cube trumps them all with a mind-blowing 7.4 quattuordecillion combinations 1. For example, a simple 2x2 Rubik's cube has over 3.5 million possible permutations. A larger cube with more individual pieces has a greater tendency to become disordered, simply because there are so many more ways that it can be arranged. If you shut your eyes and twist the sides around blindly once more, the odds of chancing upon a solved cube when you open them again are even slimmer - it is extremely unlikely that your cube will have anything but a totally random, disordered configuration.

This complex cube has many more moving parts than the first, and so has more possible permutations. With each side you twist, the cube tends towards greater disorder.StudySmarter Originals Each time you randomly twist the cube, you increase the number of possible configurations that your cube could take, decrease the chance of landing upon that perfectly solved arrangement, and get more and more disordered. But it is more likely that you will rotate a different side and disrupt the order even more. The second time you twist it, you might undo your first move and restore the cube to its original, perfectly solved arrangement. The first time you twist it, you disrupt the order. It starts off ordered - each face contains just one colour. However, when you break them down, they start to make a little more sense. In the introduction to this article, we gave you one definition of entropy.Įntropy (S) is the number of possible ways that particles and their energy can be distributed in a system.
ENTROPY EQUATION FREE
You'll find out how entropy, enthalpy, and temperature determine the feasibility of a reaction through a value known as G ibbs free energy. Finally, we'll explore the second law of thermodynamics and feasible reactions.We'll then look at entropy changes, and you'll be able to practice calculating enthalpy changes of reaction.
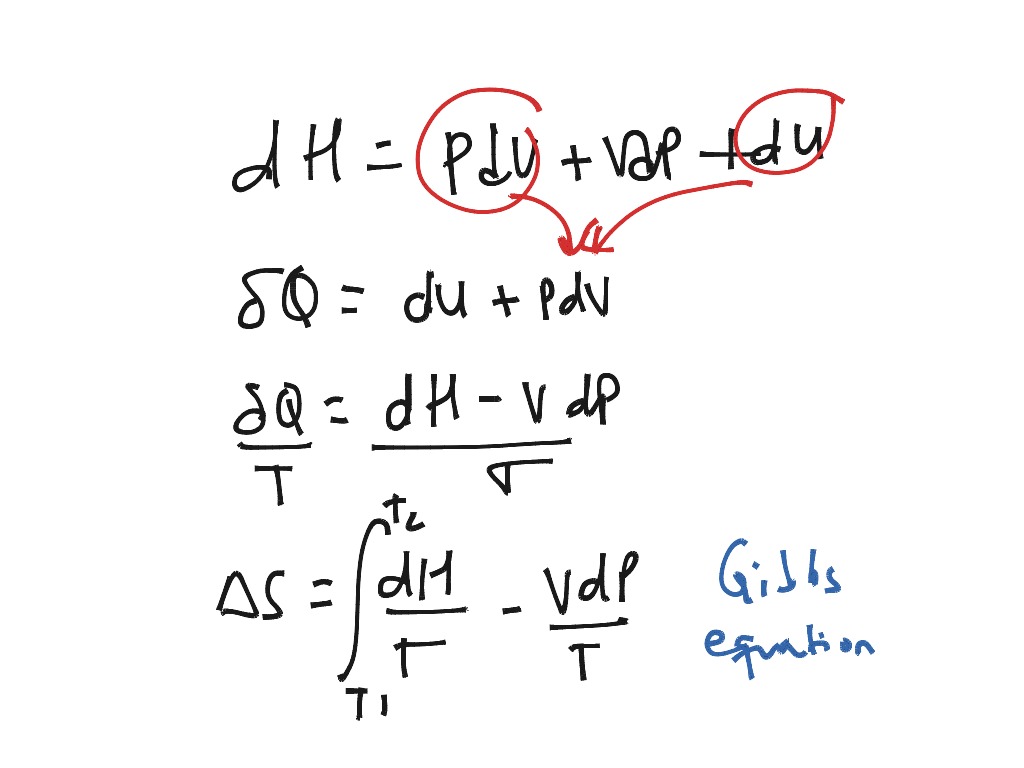
We'll start by learning the definition of entropy and its units.This article is about entropy in physical chemistry.This idea of a neat arrangement spreading out into total chaos is a good starting point for entropy: a measure of disorder in a thermodynamic system. Under random action, you could say that the faces of the cube have gone from ordered and exact to a random configuration. What are the chances that it is still perfectly solved after twisting it around blindly for a couple of minutes? They're pretty low! Instead, it is quite likely that your cube isn't perfectly solved - the faces all contain a mixture of different colours. The cube could now have all sorts of possible arrangements. Take it into your hands, shut your eyes, and twist the sides around randomly a few times. Imagine a 2x2 Rubik's cube, solved so that each face contains just one colour.

